
Ln (3/1) = (59110 / 8.31447) (1/298 minus 1/x)Ĭlicking this link will take you to a NIST paper that has a table of calculated mercury vapor pressures. Wy can I do this? Because I will be using a ratio of P 2 to P 1. I just care that it triples in value from P 1 to P 2. The enthalpy of vaporization for mercury is 59.11 kJ/mol?Ĭomment: I don't care what the actual vapor presssure value is at either temperature. By how many degrees should we increase the temperature of the flask to triple the mercury vapor pressure. The system is at room temperature of 25.0 ☌. Problem #8: A 5.00 L flask contains 3.00 g of mercury. Interestingly, Wikipedia uses the 31.4 value. The author of the above problem (not the ChemTeam!) obviously used the fourth of the four listed values. You might be interested in a collection (from the literature) of enthalpy of vaporization values for chloroform. Ln (197 / 448) = - (x / 8.31447) (1/296 minus 1/318)ģ) Let us use the Clausius-Clapeyron Equation:Ĥ) Set up equation to solve for the normal boiling point: Estimate its heat of vaporization and normal boiling point.Ģ) Set up equation to solve for the enthalpy of vaporization: Problem #7: Chloroform, CHCl 3has a vapor pressure of 197 mmHg at 23.0 ☌, and 448 mmHg at 45.0 ☌. Calculate its boiling point at 1.5 atmosphere. Problem #6: The normal boiling point of Argon is 83.8 K and its latent heat of vaporization is 1.21 kJ/mol. I just decded to walk on the wild side for a moment. That puts x in the numerator and a 1.00 in the denominator, making my calculation a bit easier. I assigned the unknown value to be associated with P 2. Problem #5: What is the vapor pressure of benzene at 25.5 ☌? The normal boiling point of benzene is 80.1 ☌ and its molar heat of vaporization is 30.8 kJ/mol. Not indicating the above in this type of question is common. This is because, by definition, the vapor pressure of a substance at its normal boiling point is 760 mmHg. What is the vapor pressure of hexane at 25.00 ☌? Problem #4: The molar enthalpy of vaporization of hexane (C 6H 14) is 28.9 kJ/mol, and its normal boiling point is 68.73 ☌.
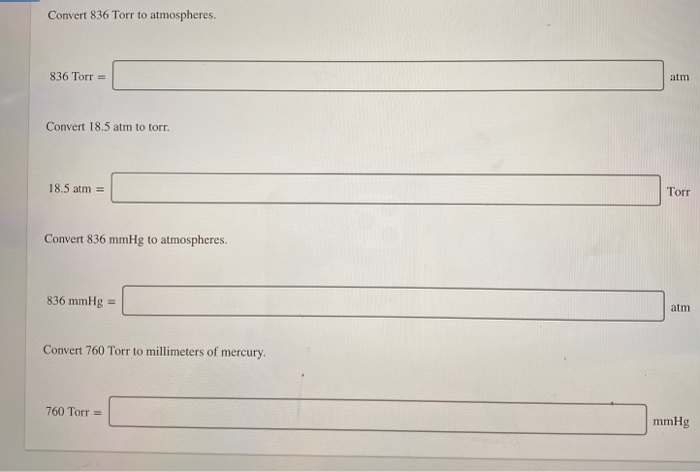
What s the enthalpy of vaporization in kJ/mol?ġ) Let us rearrange the Clausius-Clapeyron Equation: Problem #3: Carbon tetrachloride has a vapor pressure of 213 torr at 40.0 ☌ and 836 torr at 80.0 ☌. If this liquid has a normal boiling point of 105 ☌, what is the liquid's heat of vaporization in kJ/mol? Problem #2: A certain liquid has a vapor pressure of 6.91 mmHg at 0 ☌. Problem #1: Determine ΔH vap for a compound that has a measured vapor pressure of 24.3 torr at 273 K and 135 torr at 325 K.ġ) Let us use the Clausius-Clapeyron Equation: If you want more on this equation please see here. This then makes the right-hand side unitless.īelow, I plan to just solve a few problems.

The unit on R is J mol¯ 1 K¯ 1.ģ) The K¯ 1 in the temperature term will cancel with the K¯ 1 associated with R.Ĥ) This means that the unit on ΔH must be J/mol. It does not matter what units of pressure you use the only restriction is that P 1 and P 2 must be expressed using the same pressure unit.Ģ) The unit on the temperature term will be K¯ 1. Understanding means seeing that the same thing said different ways is the same thing.ġ) The natural log term on the left-hand side is unitless. I would like for you to see that they are the same equation. There is a decent chance that it will be different from the form you learned. Why do I do this? As you encounter different presentations, you will probably see whatever form of the equation the instructor (or the textbook writer) learned. I plan to use slighty different formulations of the Clausius-Clapeyron Equation in the first several questions.
